Summary – Anomeric Carbon vs Chiral CarbonĪnomeric compounds and chiral compounds are two different forms of organic compounds that have specific types of carbon atoms. The below infographic presents the differences between anomeric carbon and chiral carbon in tabular form for side by side comparison. The key difference between anomeric carbon and chiral carbon is that an anomeric carbon atom essentially contains a hydroxyl group that is either cis or trans to the exocyclic oxygen atom, whereas a chiral carbon atom essentially contains four different functional groups attached to it with single covalent bonds. What is the Difference Between Anomeric Carbon and Chiral Carbon?Īnomeric compounds and chiral compounds are two different forms of organic compounds that have specific types of carbon atoms. Since there are no chiral centres in achiral compounds, an achiral compound has superimposable mirror images. However, it has a non-superimposable mirror image. Therefore, a chiral compound has no symmetry. On the other hand, the term achiral means that there are no chiral centres present. 
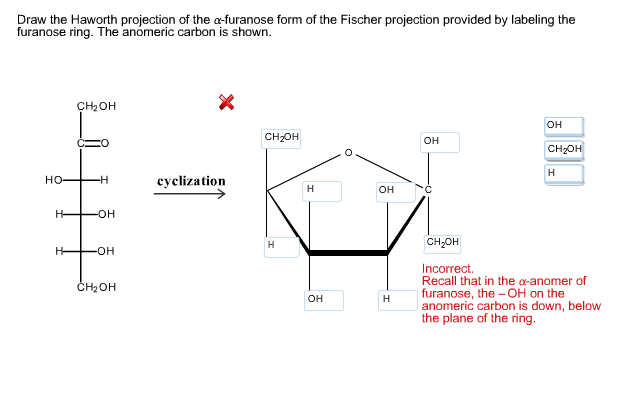
Together, these two molecules are known as enantiomers. Therefore, compounds having the chiral centre and the molecule that resembles its mirror image are two different compounds. In other words, compounds having chiral centres do not superimpose with their mirror image. The chiral centre is essentially sp3 hybridized because it has to bear four different groups of atoms, forming four single covalent bonds.įigure 02: Chiral Carbon Atoms are shown with Blue Color StarsĬhiral centres cause the optical isomerism of compounds. Chirality is actually the property of having chiral centres. Chiral compounds are compounds containing chiral carbon atoms. Chiral centre is a carbon atom of an organic compound that has four different substituents attached to it. The point that determines the presence or absence of chirality in a molecule is the chiral centre of that molecule. This term is mostly used with organic compounds. Chirality refers to the property of having a superposable mirror image. The concept of the presence of a chiral carbon is best described by chirality. What is Chiral Carbon?Ĭhiral carbon atoms are carbon atoms consisting of four single bonds to four different functional groups. That means the hydroxyl group and the exocyclic oxygen atom are on the opposite sides of the molecular projection. The following example shows the alpha anomer of D-glucopyranose.īeta anomer is the configuration of a carbohydrate in which the hydroxyl group is trans to the exocyclic oxygen at the anomeric centre. When we draw a Haworth formula, the hydroxyl group is in the downward direction if it is the alpha anomer. That means the hydroxyl group and the exocyclic oxygen atom are on the same side of the molecular projection. According to the chemical structure of anomeric carbon-containing compounds, there are two types as alpha anomers and beta anomers.Īlpha anomer is the configuration of a carbohydrate in which the hydroxyl group is cis to the exocyclic oxygen at the anomeric centre. Summary – Anomeric Carbon vs Chiral Carbon What is Anomeric Carbon?Īnomeric carbon is the carbon derived from the carbonyl carbon compound of the open-chain form of the carbohydrate molecule. Anomeric Carbon vs Chiral Carbon in Tabular Formĥ. Chiral carbon atoms are carbon atoms consisting of four single bonds to four different functional groups. The key difference between anomeric carbon and chiral carbon is that an anomeric carbon atom essentially contains a hydroxyl group that is either cis or trans to the exocyclic oxygen atom, whereas a chiral carbon atom essentially contains four different functional groups attached to it with single covalent bonds.Īnomeric carbon is the carbon originated from the carbonyl carbon compound of the open-chain form of the carbohydrate molecule. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
